Why did this happen? Understanding how Congenital Lung Malformations develop
Medically reviewed and edited by Marisa Schwab, MD | Written by Emily Lake, PhD | Last updated 04/20/2026
It is still unknown exactly why or how congenital lung malformations (CLMs) develop
But many experts believe they likely arise from disruptions during fetal lung development, which begins as early as the 4th week of pregnancy.
These are random abnormalities, probably caused by errors in molecular signaling, and not caused by genetics, environmental factors, or anything a mother did or didn’t do during pregnancy.
Congenital lung malformations are not cancer
Most are not inherited conditions: they happen randomly and—as far as we know—will not be passed on.
When parents are given a congenital lung malformation (CLM) diagnosis for their child, like CPAM or BPS, one of the first questions is almost always: Did I do something to cause this?
The answer is NO. Lung malformations arise from deeply complex biological processes that take place in the earliest weeks of pregnancy, long before most people even know they’re expecting. These conditions are random. They are not caused by anything a mother did or didn’t do.¹˒²˒³
Lung development 101
To understand why lung malformations happen, it helps to understand how the lungs form in the first place. Like fetal development in general, this is an extraordinarily complex process.
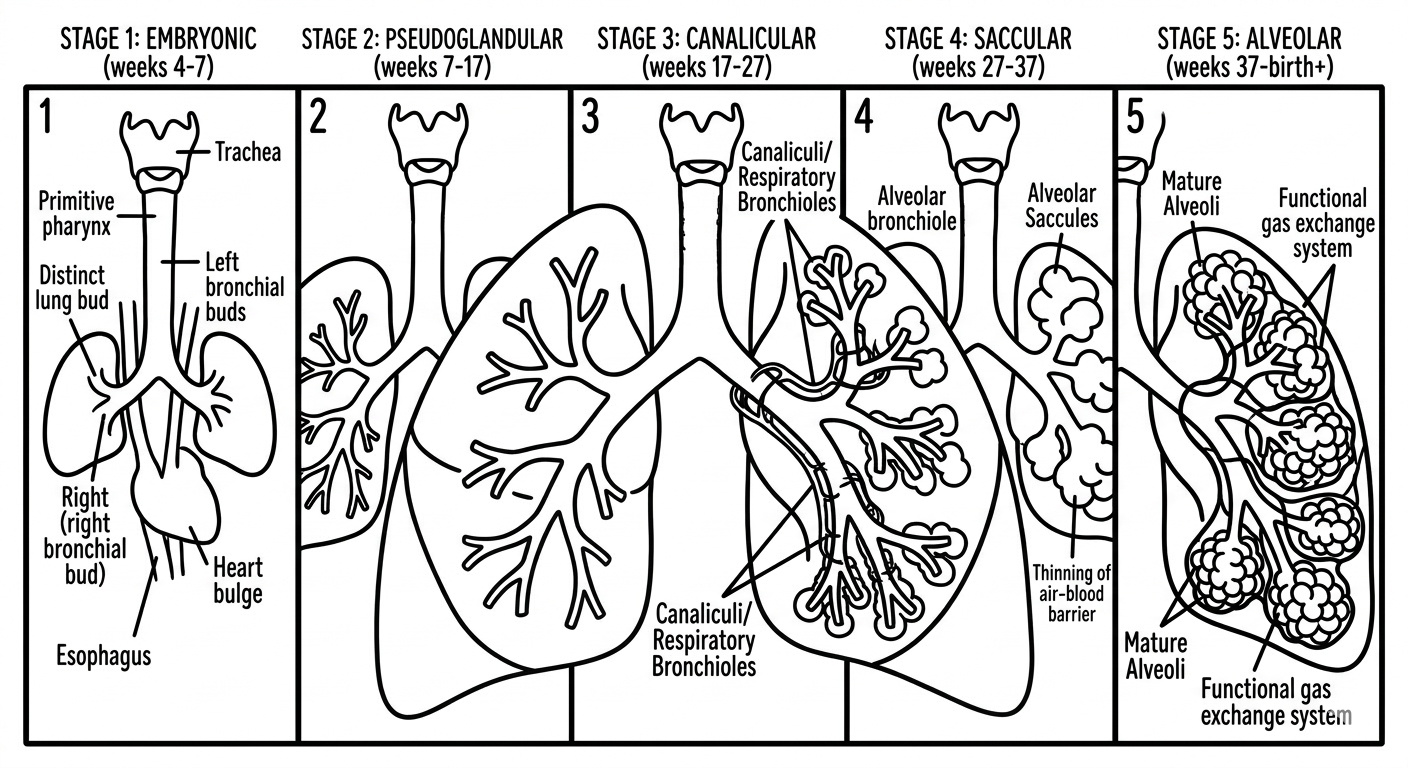
There are five overlapping stages to lung development: embryonic, pseudoglandular, canalicular, saccular, and alveolar. The lungs begin to develop extremely early in pregnancy, around the 4th week of gestation, and they continue developing well into childhood.¹˒⁴
Embryonic stage (around weeks 4–7): Two tiny buds appear. These are the very first beginnings of the lungs, branching off from the same simple tube that will become your baby’s throat and digestive tract.¹˒⁴
Pseudoglandular stage (around weeks 7–16): The lung buds branch out repeatedly, like a tree growing new limbs, forming the basic scaffolding of the future airways.¹˒⁴
Canalicular stage (around weeks 16–25): The airways start to widen and the first blood vessels grow in close alongside them, getting ready for the moment when breathing will need to begin.¹˒⁴
Saccular stage (around weeks 26–36): Tiny air sacs begin to form at the ends of the airways, and the lungs start producing surfactant, the slippery substance that keeps the air sacs from collapsing with each breath.¹˒⁴
Alveolar stage (from ~36 weeks, continuing for years after birth): The air sacs multiply and mature rapidly. Most of them actually form after birth. The lungs continue developing and growing throughout early childhood.¹˒⁴
Stages of lung development | Credit: CPAM Parents
You don’t need to understand the ins and outs of each of these stages. What’s important to take away is how intricate and complex lung development is, as well as how long the lungs keep undergoing really significant changes.
Different lung malformations are thought to reflect disruptions at different points in lung development, although these stages overlap and the exact timing is not known.¹˒²
CPAM is thought to arise when part of the lung starts growing differently very early in development.
For sequestrations, the leading theory is that a piece of lung tissue branches off independently very early in development, grows in isolation, and connects to the wrong blood supply. Sequestration means “separate”.
In congenital bronchial atresia, a small section of airway does not develop a normal connection to the rest of the bronchial tree. This leaves a dead-end blockage with trapped mucus and an over-inflated patch of lung beyond it. Atresia means that part of a tube or passage in the body did not fully form, so it ends up closed off or not connected properly.
In congenital lobar overinflation, one of the airways may be too narrow or floppy, sometimes because of abnormal cartilage development. This can act like a one-way valve: air flows in but cannot easily flow out. The result is that one section of the lung gradually becomes over-inflated, like a balloon that cannot fully deflate.¹˒²˒⁵
Signals gone (slightly) astray
Important advances in our understanding of CLMs have come from molecular biology: the study of the chemical signals that tell developing cells what to become, when to grow, and when to stop growing.¹˒³˒⁴ CLMs likely involve changes in molecular biology during lung development, like altered signaling pathways, transcription factors, or genetic alterations.
You can think of the developing lung as a construction project. The pathways are the communication system between teams on the site. The transcription factors are the site managers reading the blueprints and directing individual workers. Genetic alterations are typos in the blueprint itself. Any one of these going slightly wrong at the wrong moment can lead to one section being built differently from the original plan, even though the rest of the building remains completely unaffected.¹˒³
Signaling pathways (like SHH, WNT, BMP, FGF, and TGF-β) These are the communication networks. A pathway is like a chain of molecular events. One cell sends a signal, another receives it, and a series of effects follow.¹˒³˒⁴ If the pathway is disrupted, the resulting effect could be some kind of abnormal development, like certain lung cells not stopping growing when they should.
Transcription factors (like NKX2.1 and SOX2) These are more like site managers. They are specific proteins inside the cell’s nucleus that help decide which genes get switched on or off and when. They do not send messages between cells in the way signaling pathways do. Instead, they work inside the cell, reading the DNA and influencing what gets built next. NKX2.1 and SOX2 are two transcription factors that are particularly important in telling early lung cells what kind of cell to become. If a transcription factor is expressed at the wrong time or in the wrong amount, the cells it controls may develop incorrectly even if the incoming signals were otherwise normal.¹˒³˒⁴
Genetic alterations These are more like typos in the blueprint itself. A mutation is a change in the actual genetic code — the DNA — that alters the instructions being read.¹˒³
POSSIBLE MOLECULAR CHANGES
CPAM This is the most well-understood of the CLMs. Somatic mosaic mutations in the KRAS gene have been identified in many CPAMs and are among the most important molecular findings in the field.¹˒² KRAS normally helps regulate cell growth. When mutated it may tell cells to keep proliferating—growing—when they should stop. These are somatic mutations, meaning they happen by chance, spontaneously, in the developing lung tissue and are not inherited.¹
Altered developmental signaling, including through the FGF pathway, has also been seen in abnormal branching and cyst formation, although the exact mechanisms are still being worked out.¹˒³˒⁴ Altered expression of developmental regulators including SOX2 and TTF1/NKX2.1 has also been reported.¹˒³˒⁴
Taken together, a section of lung tissue received garbled instructions during a critical window of development and then grew abnormally.¹˒²˒³
BPS A few studies have suggested differences in developmental regulators such as HOXB5, an important gene involved in airway branching, but the molecular basis of BPS is not well understood at all.¹˒²˒³ The leading structural theory — that an extra lung bud branches off independently and connects to the wrong blood supply — is not yet proven at the molecular level.¹˒²˒⁶
CLO (Congenital Lobar Overinflation) or CLE This condition appears to be more mechanical than molecular in many cases, often involving abnormal bronchial cartilage or another structural problem affecting airflow. No specific gene mutations have been identified. In around 50% of cases no clear cause can be found at all, even after surgical examination.¹˒⁵
Bronchogenic Cysts The molecular basis of bronchogenic cysts is not yet understood. No specific gene mutations have been reliably identified.¹˒³ What we do know is that these seem to form when a small pocket grows out from the early airway as the lungs are developing. But why this happens in remains unknown.¹˒²
Bronchial Atresia Experts think this is mainly a structural problem that happens during fetal development. A small section of airway may not stay properly connected as the lungs are forming, or its blood supply may briefly be disrupted. No specific genetic or molecular cause has been found so far.
Is There a Genetic Component?
Lung malformations are not thought to be inherited conditions.¹˒² Having one child with a lung malformation does not increase the odds of it happening in a future pregnancy.¹˒² Across all CLMs, the mutations that have been found appear to be somatic: they happen randomly and are not inherited or passed on.¹˒²˒³ If your child has a family of their own one day, their children are not at higher risk of having a CLM.
Being diagnosed with a CLM is not typically a reason to need an amniocentesis because a CLM by itself is not associated with chromosomal conditions.¹˒²
In a small number of documented cases, more than one member of the same family has been found to have bronchopulmonary sequestration, and some online groups claim multiple members of the same family have had CPAM. No specific gene responsible for familial BPS or CPAM has been identified.¹˒² For the vast majority of families, a lung malformation happens as a one-off event in an individual pregnancy, with no family pattern and no known increased risk in future pregnancies.¹˒² That said, if you have a family history that feels relevant, this is absolutely worth raising with your genetic counsellor, MFM, or other specialist.
WHAT ABOUT CANCER?
Whenever genetic mutations are mentioned, many people’s minds go immediately to cancer. That is completely understandable; mutations in genes like KRAS are among the most well-known drivers of cancer in adults, and seeing them mentioned in the same breath as your baby’s diagnosis can feel really alarming. But genetic mutations are not all the same, and the story here is nuanced.¹
Not all mutations cause cancer. Mutations are simply changes in the genetic code. They happen constantly in developing tissues and most of them are inconsequential. Whether a mutation contributes to cancer depends on which gene is affected, where in the body it occurs, when it occurs during development, and many other factors we are still working to understand. Finding a cancer-associated mutation in a tissue is not the same as finding cancer, or even finding evidence that cancer will develop.¹˒³
KRAS KRAS mutations have been found in CPAM tissue.¹ If you search online, you will find KRAS comes up often in discussions of cancer, but a KRAS mutation is not automatically a “cancer mutation.” In CPAM tissue, researchers believe these mutations happened spontaneously in a small cluster of developing lung cells during fetal development.¹ They are not inherited, they are not present throughout the body, and their clinical significance in children with CPAM is still under active investigation.¹ The authors of the most comprehensive current review are explicit that this relationship requires urgent further research before conclusions can be drawn.¹
DICER1 You may also see references to a gene called DICER1 and a rare childhood lung tumor called pleuropulmonary blastoma (PPB). It is important to understand that DICER1 is not currently considered part of the usual CPAM story; it is part of a separate condition. PPB has historically been confused with CPAM because the two can look similar on imaging.¹˒² Some lesions historically labeled CPAM may in fact have always been PPB, or lesions that were difficult to distinguish from PPB using older imaging.¹˒² This is one reason older literature may overstate the cancer risk of CPAM.¹ A specific relationship between DICER1 mutations and typical CPAM has not been demonstrated.¹ PPB is also clinically a different picture: it is often symptomatic, is rarely detected before birth, and usually looks different from the kind of lesion found on a routine prenatal scan in an otherwise healthy baby.¹˒²
So what is the actual cancer risk? Honestly, we do not yet know exactly.¹ The figures that circulate online often come from studies dating back to the 1980s that used different diagnostic criteria, predated modern imaging, and likely included misclassified conditions such as PPB.¹ Current specialist estimates for prenatally diagnosed CPAMs suggest the risk is well below 1%.² The cancer question applies primarily to CPAM and hybrid lesions — congenital lobar overinflation and bronchial atresia are not thought to be associated with malignant transformation,¹ and for bronchopulmonary sequestration the limited evidence comes mostly from adult cases where lesions went undetected for many years.¹
What we don’t know yet
So much remains unknown about the full range of signaling pathways and developmental processes that underlie these abnormalities.¹˒³˒⁴ The research that has been done points firmly and consistently to the fact that these are random mutations. ¹˒²˒³ That means no lifestyle factor, environmental exposure, or maternal behavior has been identified as a cause of congenital lung malformations.¹˒²
SUMMARY
Congenital lung malformations begin very early in pregnancy, during the extremely complex process of fetal lung development. Researchers are still working to understand the exact biological mechanisms involved but the evidence so far strongly suggests that they arise randomly when a small part of the developing lung grows or forms differently from expected. Depending on the CLM, this could have a structural cause (for example, the blood supply to a small segment of developing lung tissue being temporarily disrupted) or a molecular one (for example, a change in the signals cells use to communicate during development). Although molecular changes are also discussed in cancer research, CLMs are not cancers, and the risk of a CLM becoming cancerous appears to be extremely small. For the vast majority of families a CLM is a rare, one-off event. It is not something that could have been prevented and it is not something likely to recur in a future pregnancy.
References
Pederiva F, Rothenberg SS, Hall N, et al. Congenital lung malformations. Nature Reviews Disease Primers 9, 60 (2023). https://doi.org/10.1038/s41572-023-00470-1
Kunisaki SM. Narrative review of congenital lung lesions. Translational Pediatrics 10(5):1418–1431 (2021). https://doi.org/10.21037/tp-20-133
Doktor F, Antounians L, Lacher M & Zani A. Congenital lung malformations: dysregulated lung developmental processes and altered signaling pathways. Seminars in Pediatric Surgery 31(1), 151228 (2022). https://www.sciencedirect.com/science/article/abs/pii/S1055858622000890
Saavedra B & Ortega F. Congenital Pulmonary Malformations: Classification and Pathogenesis. Medical Research Archives / European Society of Medicine (2025). https://esmed.org/MRA/mra/article/view/6775
StatPearls (NCBI Bookshelf). Congenital Pulmonary Airway Malformation. Updated 2023. https://www.ncbi.nlm.nih.gov/books/NBK551664/
Pederiva F et al. Developmental Pathways Underlying Lung Development and Congenital Lung Disorders. PMC / Frontiers (2021). https://pmc.ncbi.nlm.nih.gov/articles/PMC8616556/
Dimopoulou A et al. Congenital Lung Malformations: A Pictorial Review of Imaging Findings and a Practical Guide for Diagnosis. Children 11(6):638 (2024). https://www.mdpi.com/2227-9067/11/6/638
Johns Hopkins Medicine. Congenital Lung Malformations. https://www.hopkinsmedicine.org/health/conditions-and-diseases/congenital-lung-malformations
Peiffer S et al. Novel Clinical Algorithm for Prenatal Monitoring of Congenital Lung Malformations. Journal of Surgical Research 293:373–380 (2024). https://pubmed.ncbi.nlm.nih.gov/37806224/
Berrocal T et al. Congenital Lung Abnormalities: Embryologic Features, Prenatal Diagnosis, and Postnatal Radiologic-Pathologic Correlation. RadioGraphics (2004). https://pubs.rsna.org/doi/abs/10.1148/rg.306105508